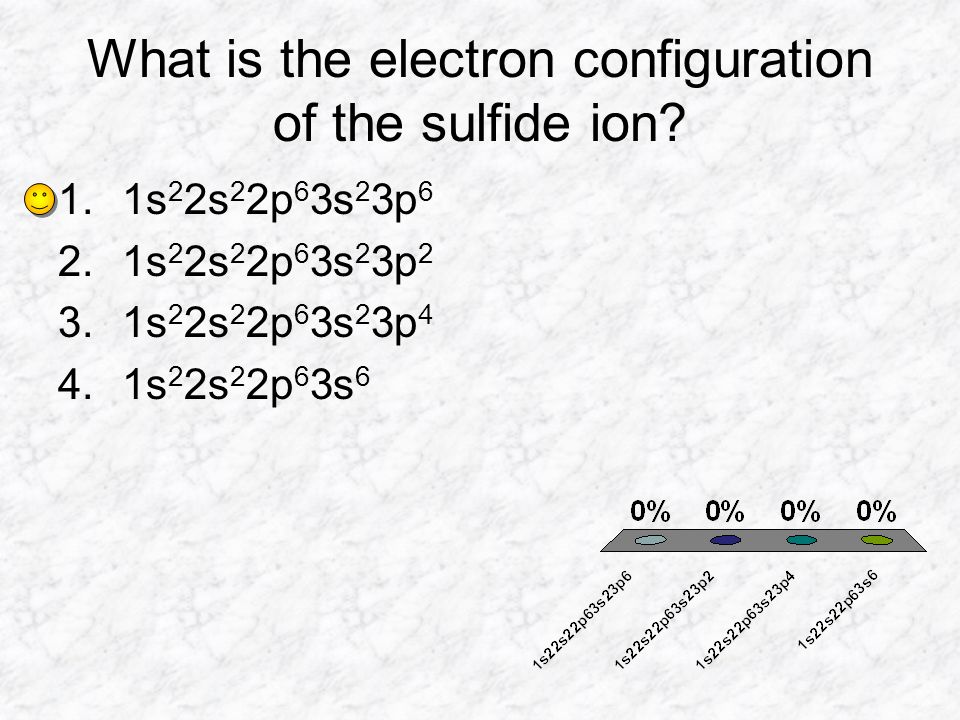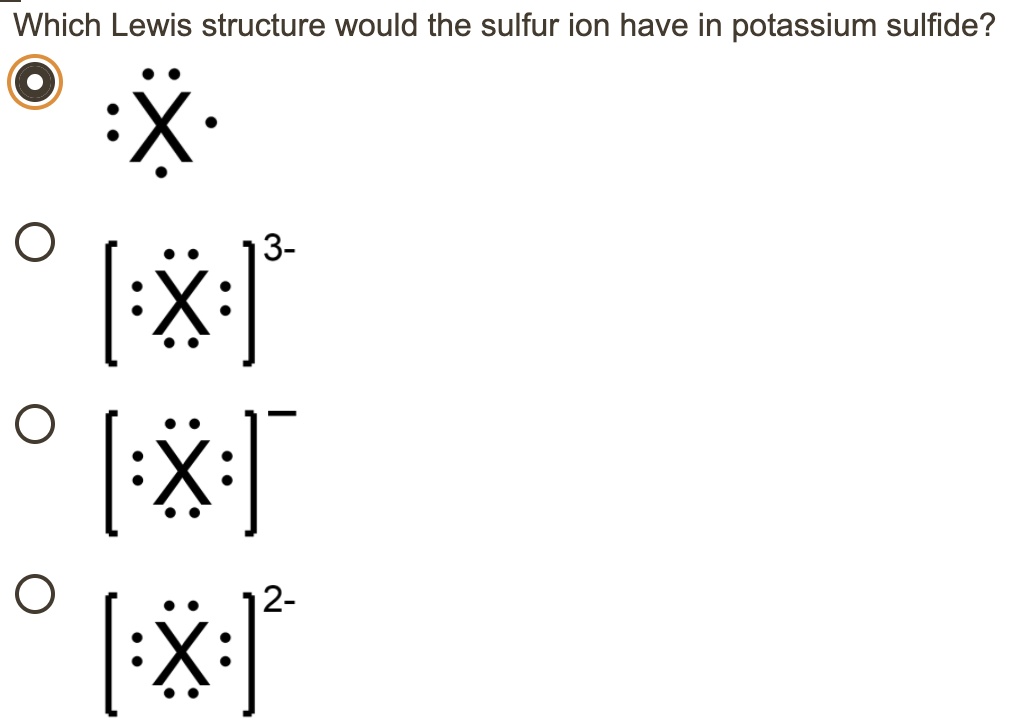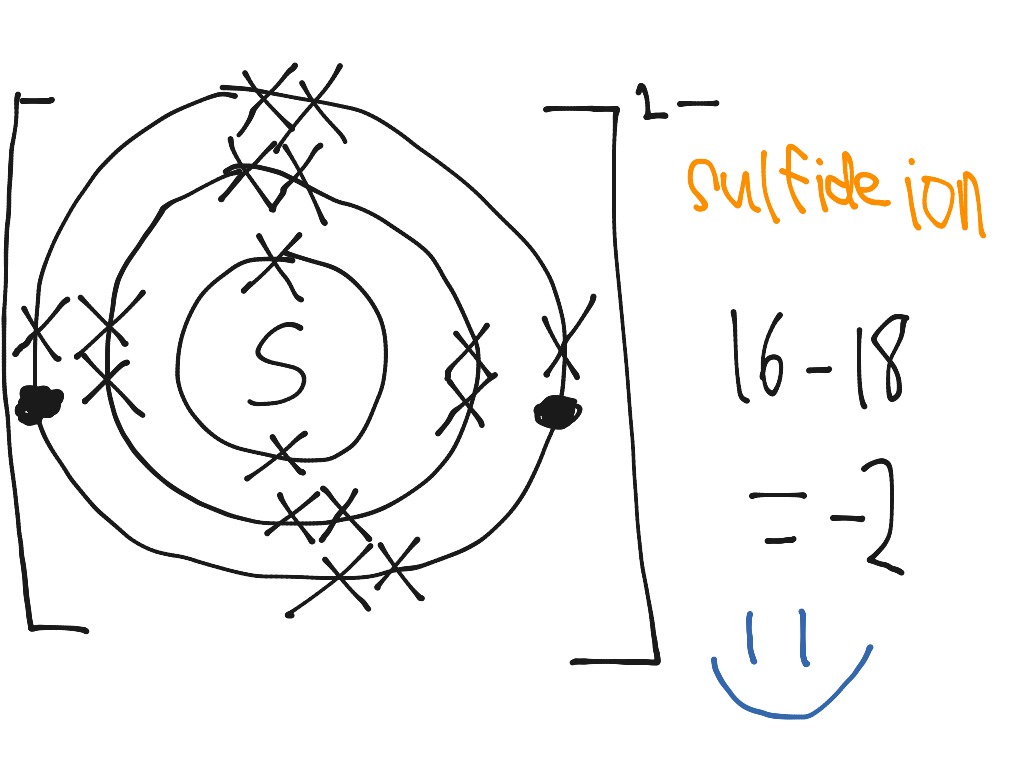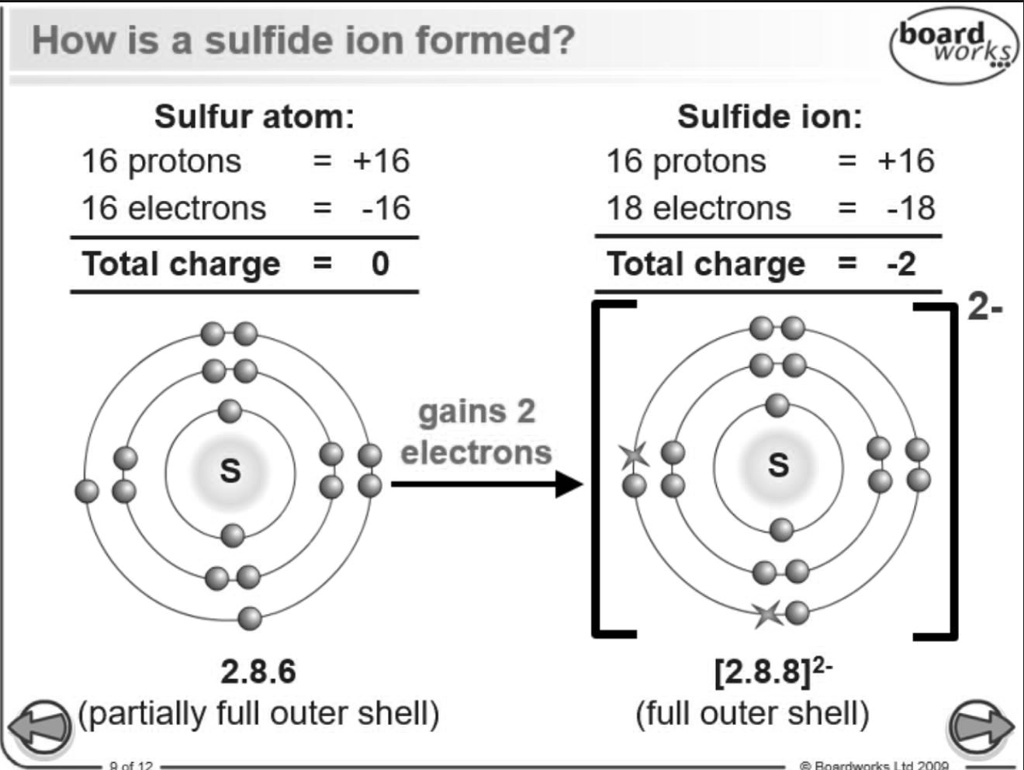
SOLVED: 'I thought in the third shell there are 18 electrons, but why are they gaining only 2 electrons?? Pls tell me... I have exam tomorrow.... How is a sulfide ion formed?

Binary Iron Sulfide as a Low-Cost and High-Performance Anode for Lithium-/Sodium-Ion Batteries | ACS Applied Materials & Interfaces

A fundamental study considering specific ion effects on the attachment of sulfide minerals to air bubbles - ScienceDirect