
Jual nah2po4 h20 sodium dihydrogen phosphate monohydrate merck 1.06346 - Kota Surabaya - mutiara bersaudara | Tokopedia

High Quality Sodium Phosphate Monobasic CAS: 13472-35-0 Nah2po4 2H2O - China High Quality Sodium Phosphate Monobasic and Ar Gradesodium Phosphate Monobasic
![Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs](https://www.bioland-sci.com/images/NaH2PO4s%20500G.jpg)
Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 500g) [CN04-500G] - $20.00 : Bioland Scientific, for Your Research Needs
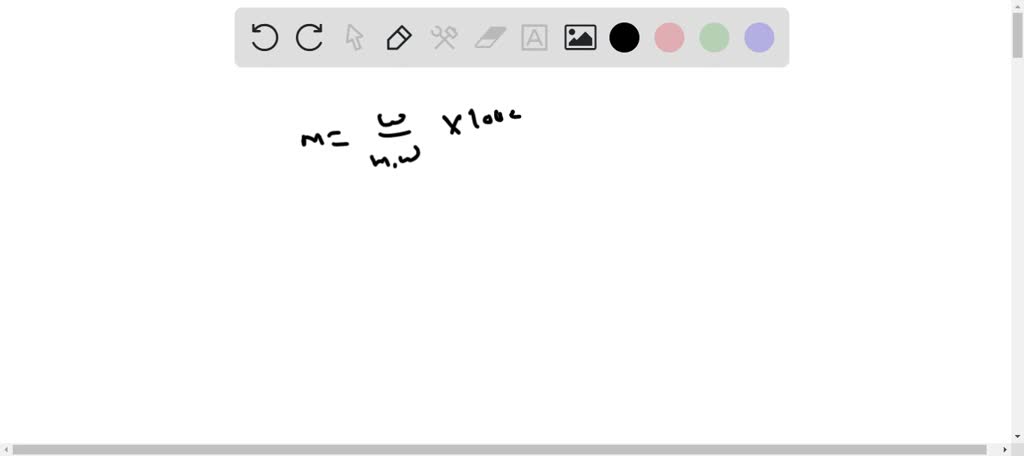
SOLVED: Weigh out 10.4 g sodium phosphate (NaH2PO4.H2O, MW = 138 g/mol). Dissolve the NaH2PO4.H2O in 450 mL water. Bring pH of solution to 7.4 with 10 M NaOH (need about 2









![PART 1: DATA [4 POINTS] Mass of NaH2PO4+H2O = 0.65 g | Chegg.com PART 1: DATA [4 POINTS] Mass of NaH2PO4+H2O = 0.65 g | Chegg.com](https://media.cheggcdn.com/media/1ae/1aecabab-9b80-48fe-99b2-8b304c594dc1/php5IPXYg)



