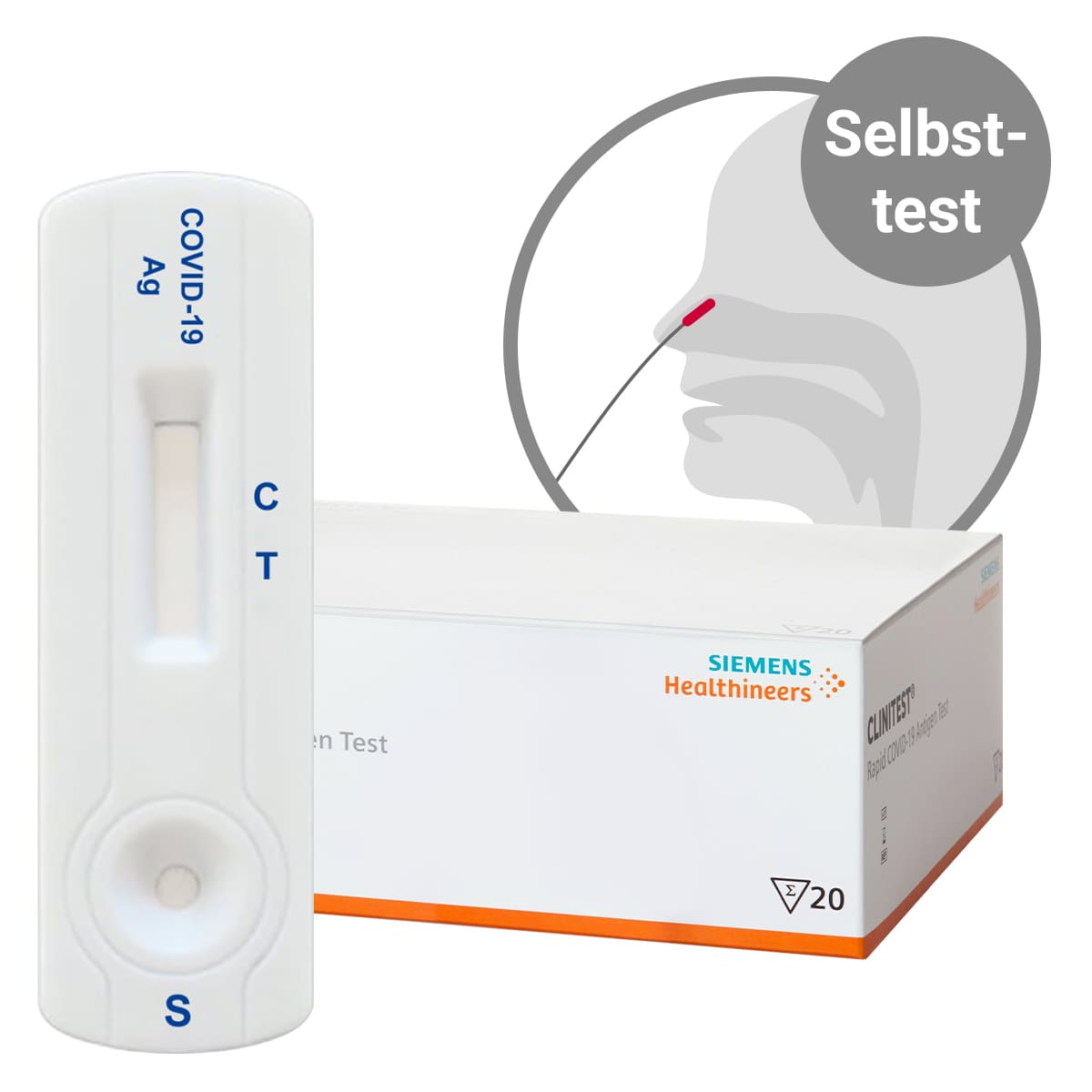
Millions of Siemens Healthineers CLINITEST Rapid COVID-19 Antigen Self-Test Now Available in the U.S. | Business Wire
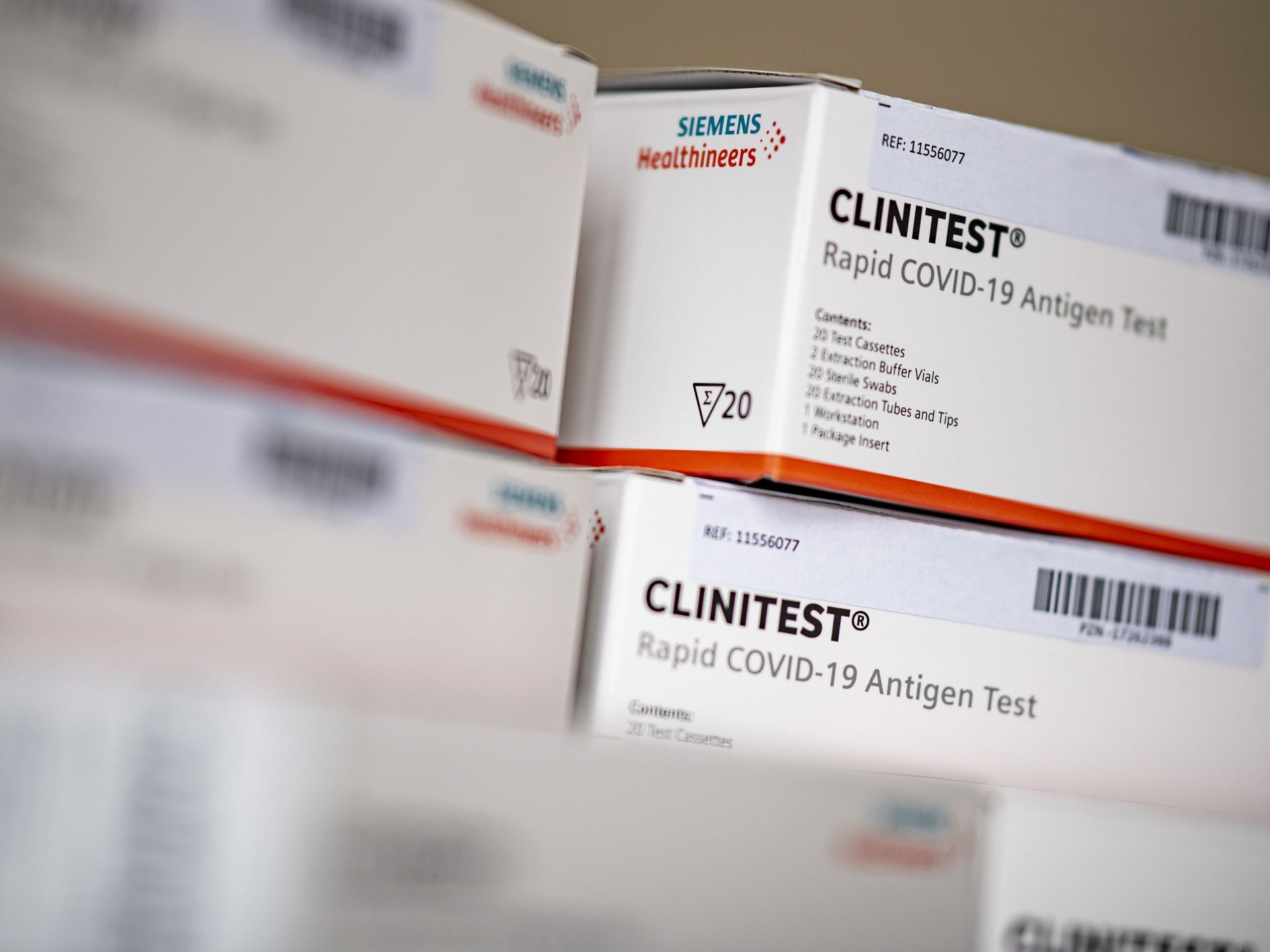
Siemens Healthineers Gains EUA For CLINITEST Rapid COVID-19 Antigen Self-Test | Medical Product Outsourcing

FDA grants emergency use nod to Siemens Healthineers' COVID-19 Rapid Antigen Self-Test | Seeking Alpha

Benjy Renton on Twitter: "And the 11th at-home test is born! Through the Biden administration's accelerated pathway to authorizing new rapid tests, the FDA has authorized Siemens Healthineers' at-home antigen test. This

CLINITEST Rapid COVID-19 Antigen Self-Test | United States of America | CLINITEST Rapid COVID-19 Antigen Self-Test has received FDA Emergency Use Authorization in the United States. https://bit.ly/3zdnMyX | By Siemens Healthineers

COVID-19 Antigen Self-Test from Siemens Healthineers receives special approval in Germany for self-administration by lay persons