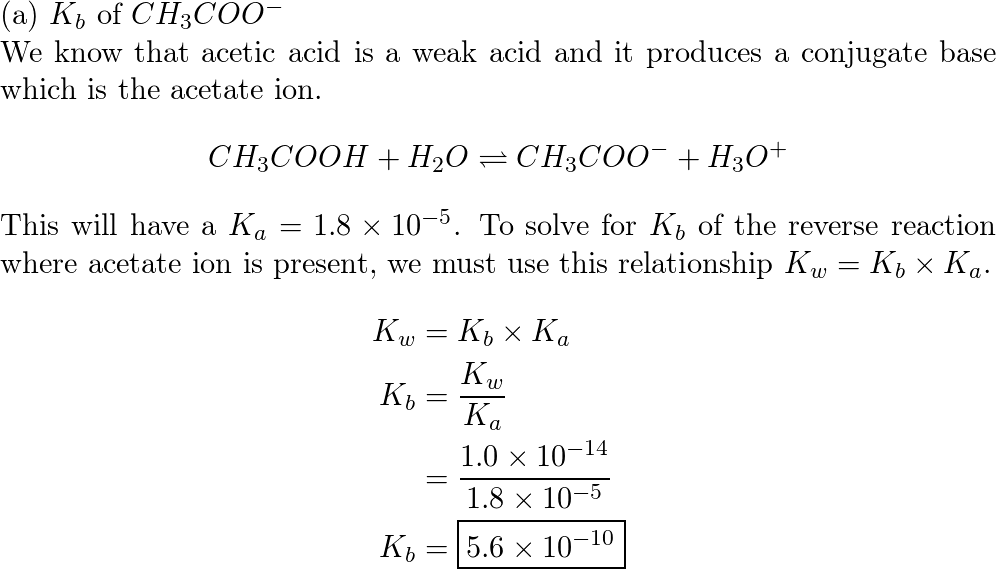Simultaneous quantitative analysis of the acetate, formate, chloride, phosphate and sulfate anions in biodiesel by ion chromatography - ScienceDirect
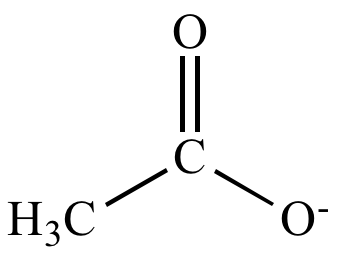
SOLVED: Determine the shape around each central atom in the acetate ion. Draw the Lewis structure first. (enter one of the following: linear, trigonal planar, tetrahedral, trigonal pyramidal, bent, trigonal bipyramidal, seesaw,
![SOLVED: Sa Acetate ion is a better leaving group than methoxide ion in both Swl and Sw2 reaction Explain why: [4 points] CHzO CH; Acetate ion Methoxide ion Sb. Circle the better SOLVED: Sa Acetate ion is a better leaving group than methoxide ion in both Swl and Sw2 reaction Explain why: [4 points] CHzO CH; Acetate ion Methoxide ion Sb. Circle the better](https://cdn.numerade.com/ask_images/75714da48f814e6d9f7e1d00f37c87d1.jpg)

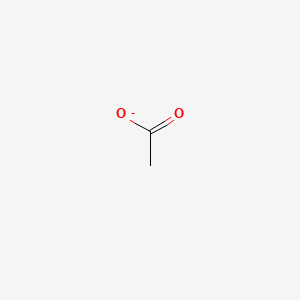

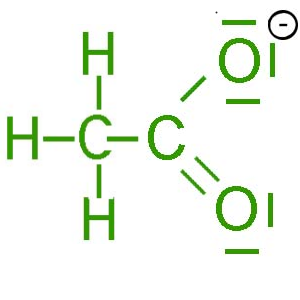
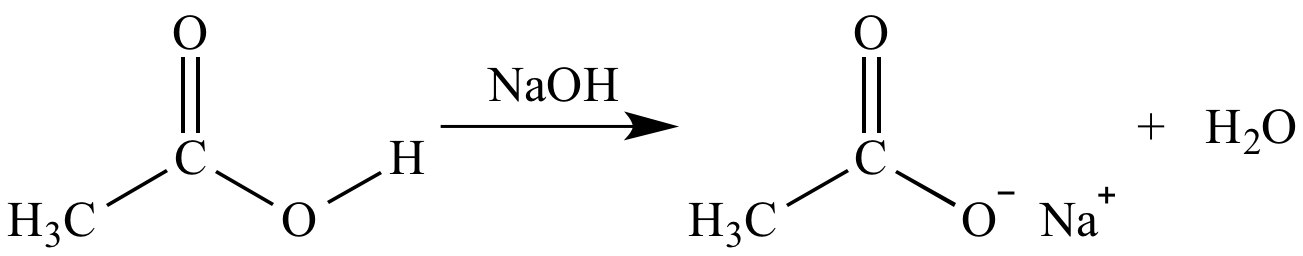



![Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion. Acetate [C2H3O2(−)] - Structure, Properties, Molecular Weight & Uses of Acetate ion.](https://cdn1.byjus.com/wp-content/uploads/2019/04/structure-of-acetate.png)