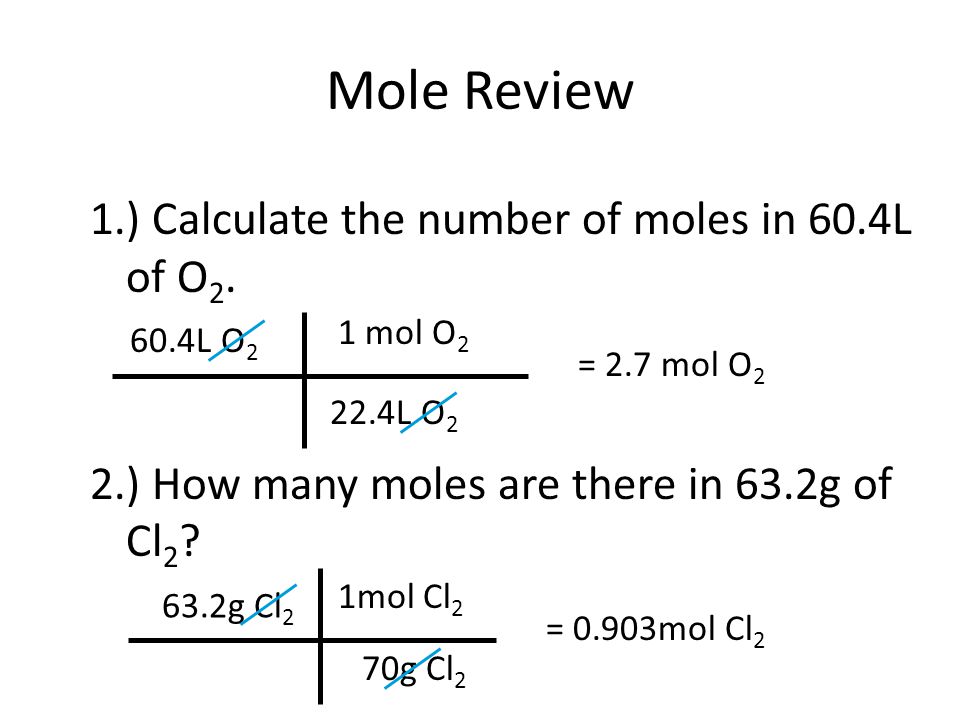
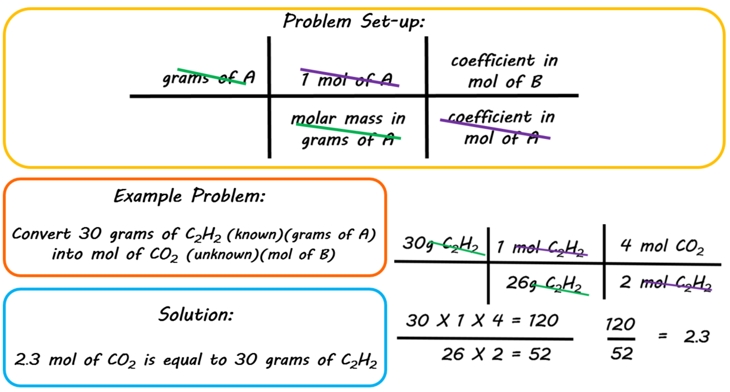
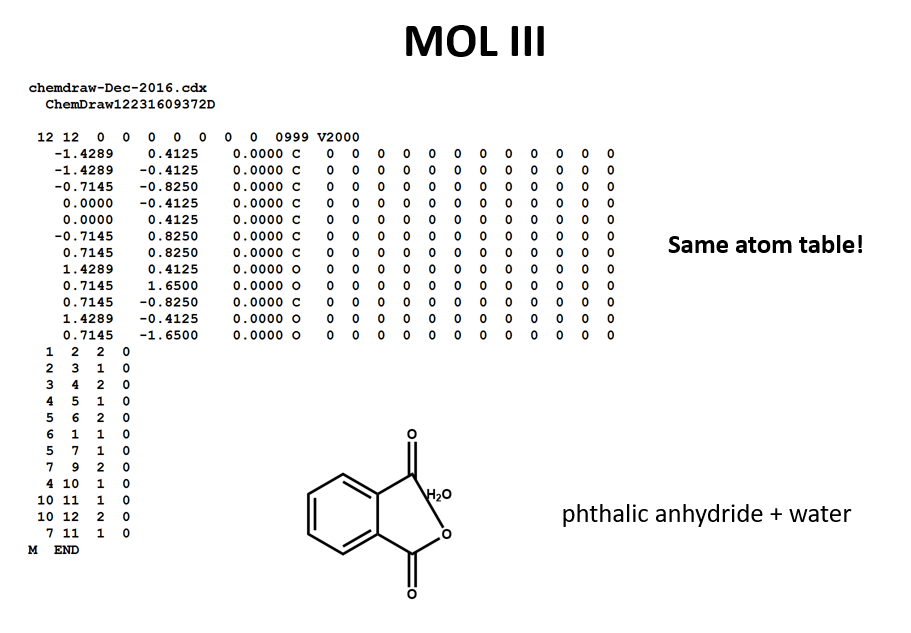
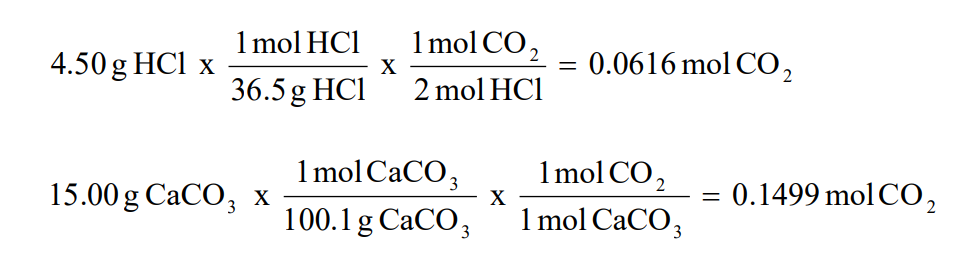2 moles of an ideal gas at temp 27 degree c is heated isoermall from volume v to 4v .if R=2 cal/mol then the heat input in the process is approximately

How to Convert from Moles of One Substance to Moles of Another Substance (Stoichiometry part 1) - YouTube

For one mole of a Van der Waals gas when b = 0 and T = 300 K, the PV vs 1V plot is shown above. The value of the Van der

SOLVED: Q9.A sample consisting of 2.00 mol He is expanded isothermally 22'C from 22.8 dm' to 31.7 dm' reversibly against constant external pressure equal to the final pressure of the gas (C)

Question Video: Calculating the Percentage by Mass of Water in Alum Given Its Chemical Formula | Nagwa

SOLVED: What is the formula weight (in g/mol) of copper(II) nitrate trihydrate, Cu(NO3)2•3H2O? Give your answer to 6 significant figures. g/mol What is the percentage of copper (by mass) in copper(II) nitrate

Fly London MOL 2 Camel - Kostenloser Versand | Spartoo.de ! - Schuhe Klassische Stiefel Damen 159,20 €

The rate constant of a reaction is 9.2 × 10 5 mol 2 L 2 s 1. The order of the reaction is:A. 3B. 2C. ZeroD. 1